Understanding the language of cosmetic ingredients is essential for anyone developing personal care products, whether you're launching your first brand or expanding an established line. INCI ingredients form the foundation of product transparency and regulatory compliance across the global beauty industry. This standardized naming system ensures that consumers, formulators, and regulatory bodies all speak the same language when discussing what goes into cosmetic products. For entrepreneurs and brands working with custom formulations, mastering INCI nomenclature isn't just about compliance-it's about building trust, ensuring safety, and creating products that meet international standards.
What Are INCI Ingredients and Why They Matter
The International Nomenclature of Cosmetic Ingredients (INCI) represents a systematic method for naming cosmetic ingredients that's recognized worldwide. Developed to create consistency across borders and languages, this nomenclature system assigns a unique, standardized name to every ingredient used in cosmetic and personal care formulations.
When you examine a product label, those scientific-sounding names aren't randomly chosen. Each INCI name follows specific conventions established by the International Cosmetic Ingredient Nomenclature Committee, ensuring that "Aqua" means water whether you're manufacturing in California or selling in Paris. This universal approach eliminates confusion and protects consumers by making ingredient lists transparent and comparable across brands.
The History and Development of INCI Standards
The INCI system emerged from a critical need for standardization in an increasingly global marketplace. Before this system existed, the same ingredient might appear under different names in different countries, creating confusion for consumers and regulatory challenges for manufacturers.
The Personal Care Products Council established the framework that would become today's INCI standards. This organization recognized that as cosmetic products crossed borders with increasing frequency, a unified naming convention would benefit everyone from formulators to end users. The system they created balances scientific accuracy with practical application, using primarily Latin botanical names for plant-derived ingredients and systematic chemical nomenclature for synthetic compounds.
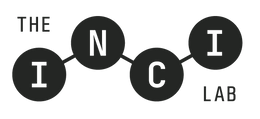
How INCI Ingredients Are Named and Classified
Understanding how ingredients receive their INCI designations helps formulators make informed decisions when developing personal care products. The naming process follows strict protocols that consider the ingredient's source, chemical structure, and function.
Botanical ingredients receive names based on their Latin binomial nomenclature, followed by the part of the plant used. For example, chamomile extract appears as "Chamomilla Recutita (Matricaria) Flower Extract," indicating both the species and the specific plant component. This precision ensures that formulators know exactly which botanical they're working with, as different parts of the same plant can have vastly different properties.
Chemical compounds follow systematic chemical nomenclature, though common names are used when they're well-established in scientific literature. Vitamin C might appear as "Ascorbic Acid," while more complex molecules receive longer, more technical designations that reflect their chemical structure.
Reading and Interpreting INCI Ingredient Lists
The order of inci ingredients on a product label isn't arbitrary-it follows regulatory requirements that provide valuable information about formulation composition. Ingredients must be listed in descending order of predominance, meaning the first ingredient listed is present in the highest concentration.
This ordering system continues until you reach the 1% threshold. Below this concentration, ingredients can be listed in any order, which is why you'll often see preservatives and fragrances clustered at the end of ingredient lists regardless of their exact percentages.
| Concentration Range | Listing Requirement | Typical Ingredients |
|---|---|---|
| Above 1% | Descending order by weight | Water, primary actives, emollients |
| Below 1% | Any order | Preservatives, fragrances, colorants |
| Trace amounts | Must still be listed | Processing aids, residual materials |
Understanding this structure helps both formulators and consumers make informed decisions. When you see "Aqua" or "Water" as the first ingredient, you know it comprises the largest portion of the formulation. This transparency is exactly what the INCI system was designed to provide.
INCI Ingredients in Custom Formulation Development
For brands working on custom cosmetic formulations, mastering INCI nomenclature becomes a strategic advantage. Every ingredient selection affects not just product performance but also label appeal, regulatory compliance, and market positioning.
When developing new products, formulators must consider how INCI names will appear to target consumers. Some ingredients have INCI names that sound harsh or chemical, even when they're derived from natural sources. "Sodium Hydroxide" might alarm some consumers, even though this ingredient is essential for pH adjustment in many formulations and is completely neutralized in the final product.
Navigating Natural vs. Synthetic INCI Designations
The distinction between natural and synthetic ingredients isn't always clear from INCI names alone. An ingredient like "Tocopherol" (Vitamin E) could be naturally derived from vegetable oils or synthetically produced in a laboratory-the INCI name doesn't specify the source.
This is where understanding personal care ingredients becomes crucial for brand development. Formulators must maintain detailed specifications that go beyond INCI names to include:
- Source and origin of raw materials
- Processing methods and refinement techniques
- Purity grades and quality standards
- Sustainability certifications and ethical sourcing documentation
Brands focused on eco-friendly cosmetic practices need this additional layer of information to ensure their ingredients align with their values and marketing claims.
Common INCI Ingredients and Their Functions
Certain inci ingredients appear frequently across product categories because of their proven effectiveness and safety profiles. Recognizing these common ingredients helps streamline formulation decisions and regulatory submissions.
Water and Aqueous Bases
Aqua appears first on most cosmetic ingredient lists, serving as the primary solvent and delivery system for active ingredients. While it might seem simple, the quality and treatment of water in formulations significantly impacts product stability and performance.
Distilled or deionized water ensures purity and prevents unwanted mineral interactions with other ingredients. Some formulations use floral waters or herbal infusions, which would be listed by their specific INCI names like "Rosa Damascena Flower Water."
Emollients and Moisturizers
These ingredients create the sensory experience of cosmetic products while delivering functional benefits:
- Glycerin: A powerful humectant that attracts moisture to the skin
- Cetearyl Alcohol: A fatty alcohol that provides conditioning and emulsion stability
- Dimethicone: A silicone that creates smooth texture and water resistance
- Butyrospermum Parkii (Shea) Butter: A natural emollient rich in fatty acids
Active Ingredients and Performance Compounds
Active ingredients drive product claims and consumer interest. Their INCI names often become marketing focal points:
- Niacinamide: Vitamin B3, known for brightening and barrier support
- Hyaluronic Acid: Actually listed as "Sodium Hyaluronate" in most formulations
- Retinol: A vitamin A derivative for anti-aging applications
- Ascorbic Acid: The most common form of vitamin C in skincare
Regulatory Requirements and INCI Compliance
Compliance with INCI naming conventions isn't optional-it's a legal requirement in most markets. The United States, European Union, and many other jurisdictions mandate proper INCI labeling for cosmetic products sold within their borders.
The INCI nomenclature conventions provide detailed guidance on proper ingredient declaration. These conventions cover everything from standard naming protocols to special situations involving mixtures, complexes, and proprietary blends.
Obtaining and Verifying INCI Names
When working with new or unusual ingredients, formulators need to verify the correct INCI designation. The process typically involves:
- Consulting official INCI databases and resources
- Reviewing supplier documentation and certificates of analysis
- Confirming names with the International Cosmetic Ingredient Nomenclature Committee
- Cross-referencing with regulatory databases in target markets
Many ingredient suppliers provide INCI names in their technical documentation, but it's crucial to verify these against official sources. Incorrect INCI names on product labels can result in regulatory violations, product recalls, and damage to brand reputation.
| Verification Method | Reliability | Best Used For |
|---|---|---|
| Official INCI database | Highest | All ingredients |
| Supplier documentation | High (if reputable) | Common ingredients |
| Cosmetic ingredient resources | Medium to High | Research and confirmation |
| Ingredient decoder tools | Medium | Consumer education |
INCI Ingredients and Marketing Strategy
The way inci ingredients appear on labels directly impacts consumer perception and purchasing decisions. Modern consumers increasingly scrutinize ingredient lists, using apps and databases to decode ingredient lists and research product composition.
Brands must balance regulatory compliance with marketing appeal. This means making strategic formulation choices that result in ingredient lists that satisfy both legal requirements and consumer preferences.
Clean Beauty and INCI Transparency
The clean beauty movement has placed unprecedented focus on ingredient transparency. Consumers want to understand not just what's in their products, but why each ingredient is included and where it comes from.
This trend has created opportunities for brands that can communicate effectively about their ingredient choices. Rather than hiding behind complex INCI names, successful brands embrace them as opportunities for education and differentiation.
Strategies for effective INCI communication include:
- Creating ingredient glossaries that explain INCI names in consumer-friendly language
- Highlighting star ingredients and their benefits prominently on packaging
- Providing detailed information about sourcing and sustainability for key ingredients
- Being transparent about the function of preservatives and stabilizers
When launching a new skincare brand, your ingredient story becomes a crucial differentiator in crowded markets.

Advanced INCI Considerations for Formulators
Professional formulators deal with complex INCI scenarios that go beyond basic ingredient listing. These situations require deeper understanding of nomenclature rules and regulatory nuances.
Ingredient Complexes and Mixtures
When ingredients are supplied as pre-mixed complexes, the INCI declaration must reflect all components. A botanical extract might contain the plant material plus the extraction solvent and any stabilizers added by the supplier. Each component must be listed separately unless it qualifies as an established mixture with its own INCI designation.
Common complex situations include:
- Preserved ingredient systems requiring listing of preservatives
- Extracts with carrier oils or solvents
- Encapsulated or complexed active ingredients
- Trade name materials containing multiple components
The INCI naming committee provides guidance for these scenarios, but formulators often need to work closely with suppliers to ensure accurate declarations.
Trade Names vs. INCI Names
Ingredient suppliers often market materials under trade names that bear little resemblance to their INCI designations. A supplier might sell "SuperHydra Complex," but the actual INCI listing could include five or more individual ingredients.
Formulators must maintain detailed documentation that connects trade names to INCI names. This documentation becomes essential during:
- Label development and artwork approval
- Regulatory submissions and safety assessments
- Manufacturing scale-up and raw material procurement
- Quality control and batch record keeping
Understanding this distinction helps prevent costly labeling errors and regulatory complications.
INCI Ingredients and Global Market Access
Different markets have varying requirements for how inci ingredients must be declared and documented. While the INCI system provides standardization, regional regulations add layers of complexity for brands selling internationally.
Regional Variations in INCI Requirements
The European Union requires INCI names as established by the EU's CosIng database, which may differ slightly from INCI names used in other regions. China has specific requirements for ingredient registration and declaration that go beyond standard INCI listings. The United States follows FDA regulations that reference but don't always mandate INCI nomenclature.
Key regional considerations:
- EU/UK: Mandatory INCI use, specific allergen labeling requirements
- USA: INCI recommended but not always required, FDA oversight
- China: INCI with additional local name translations required
- Japan: INCI accepted alongside local ingredient designations
Brands planning international expansion need to understand these nuances from the formulation stage. Choosing ingredients with clear regulatory status in target markets simplifies the path to market access.
Documentation and Substantiation
Proper INCI documentation extends beyond simple ingredient lists. Comprehensive formulation records should include:
| Documentation Type | Purpose | Regulatory Relevance |
|---|---|---|
| Full INCI listing | Product labeling | Required in most markets |
| Raw material specifications | Quality control | Safety assessments |
| Certificates of analysis | Batch verification | Audit requirements |
| Allergen declarations | Consumer safety | EU mandatory labeling |
| Function statements | Claim substantiation | Marketing compliance |
This documentation supports not just regulatory compliance but also quality management and brand protection strategies.
Future Trends in INCI Standards and Applications
The INCI system continues to evolve as new ingredients enter the market and consumer expectations shift. Understanding these trends helps brands stay ahead of regulatory changes and market demands.
Biotechnology and Novel Ingredients
Biotechnology is producing innovative ingredients that challenge traditional INCI naming conventions. Fermentation-derived actives, bioengineered peptides, and lab-grown botanicals require new approaches to nomenclature and classification.
The INCI committee regularly updates its guidance to accommodate these innovations while maintaining the system's core principles of clarity and consistency. Formulators working with cutting-edge ingredients should monitor these updates and engage with suppliers to ensure proper ingredient declaration.
Sustainability and Traceability
Consumers increasingly demand information about ingredient sustainability that goes beyond INCI names. While INCI provides standardization, it doesn't convey information about farming practices, carbon footprint, or ethical sourcing.
Forward-thinking brands are supplementing INCI listings with additional transparency measures such as:
- QR codes linking to detailed ingredient sourcing information
- Sustainability certifications displayed alongside INCI names
- Digital platforms providing deep-dive ingredient education
- Blockchain-based traceability systems for key ingredients
These approaches build consumer trust while maintaining INCI compliance as the regulatory foundation.
Practical INCI Resources for Brand Development
Access to reliable INCI resources streamlines formulation development and regulatory compliance. Professional formulators maintain libraries of reference materials and databases to support their work.
Essential INCI Reference Tools
The cosmetic ingredient dictionary provides searchable databases of INCI names with functional descriptions and usage guidance. These resources help formulators quickly identify appropriate ingredients and understand their regulatory status.
Additional valuable resources include:
- Official INCI nomenclature manuals updated annually
- Supplier technical documentation with complete INCI listings
- Regulatory databases specific to target markets
- Professional formulation software with built-in INCI libraries
For brands working with formulation partners, ensuring your partner maintains current INCI resources is essential. The formulation expertise required for compliant product development depends on access to accurate, up-to-date ingredient information.
Working with Formulation Partners
Professional formulation labs maintain comprehensive INCI databases and regulatory expertise that individual brands often lack. When evaluating potential formulation partners, consider their capabilities in:
- INCI compliance and documentation: Complete, accurate ingredient listings
- Regulatory knowledge: Understanding of regional requirements
- Ingredient sourcing: Access to properly documented raw materials
- Quality systems: Processes ensuring INCI accuracy across batches
The right formulation partner transforms INCI compliance from a challenge into a competitive advantage, creating products that meet regulatory standards while delivering on brand promises.
Mastering INCI ingredients is fundamental to success in cosmetic product development, enabling regulatory compliance, consumer trust, and market access across global territories. Whether you're launching your first product or expanding an established line, understanding this universal language of cosmetic ingredients ensures your formulations meet professional standards and consumer expectations. The INCI Lab combines deep expertise in ingredient selection and INCI compliance with award-winning formulation capabilities, providing entrepreneurs and established brands with custom solutions that deliver both performance and transparency. From concept through production, our team ensures your products stand out with formulations that are properly documented, sustainably sourced, and designed for success in competitive markets.