The beauty and personal care industry operates under rigorous regulatory frameworks designed to protect consumers while enabling innovation. For brands developing products in the Oceania region, understanding skincare formulation standards in New Zealand and Australia is essential for market entry and long-term success. These two neighboring countries maintain distinct yet interconnected regulatory systems that govern ingredient safety, manufacturing practices, labeling requirements, and quality assurance protocols. Whether you're launching a new cosmetic brand or expanding your existing product line, navigating these standards requires comprehensive knowledge of both countries' regulatory environments and a commitment to excellence in formulation development.
Understanding the Regulatory Framework
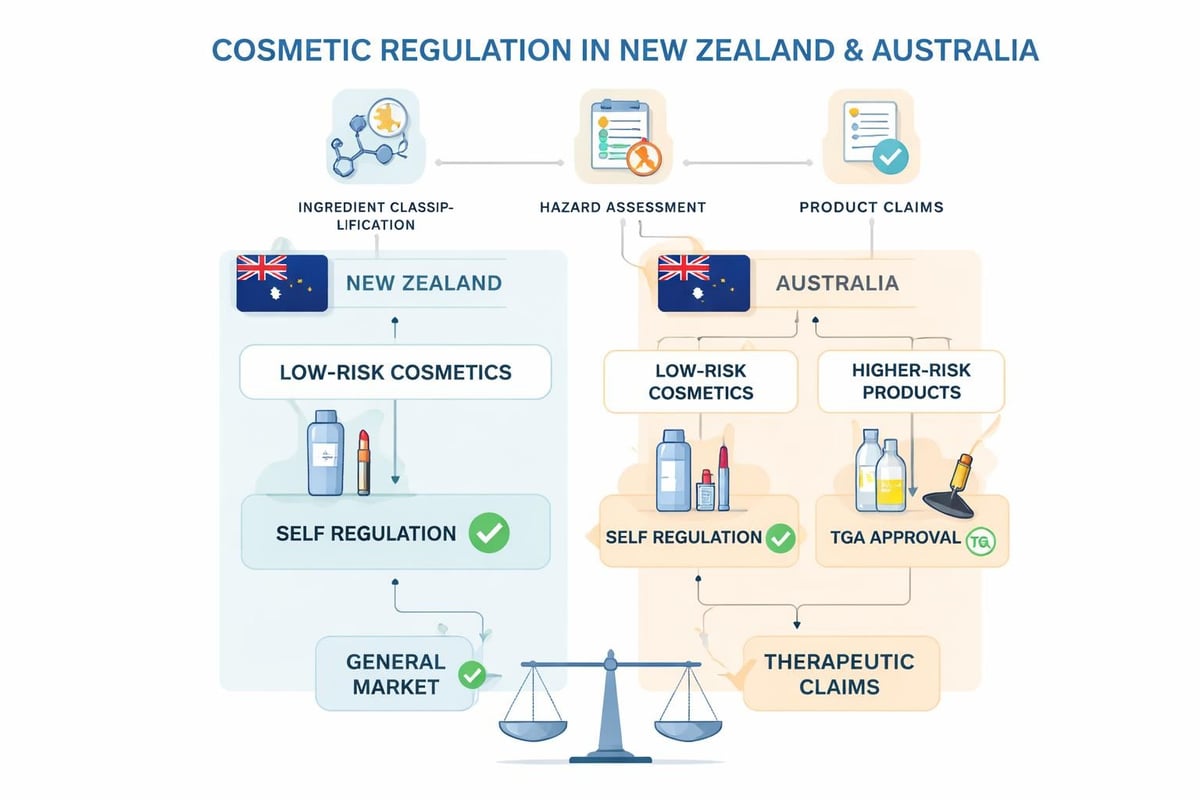
The regulatory landscape for cosmetics in New Zealand and Australia follows different pathways despite the countries' close economic relationship. Australia maintains one of the most structured cosmetic regulatory systems globally through the Industrial Chemicals Act 2019, which requires notification of cosmetic products and their ingredients. The Australian system emphasizes pre-market assessment and ongoing compliance monitoring.
New Zealand takes a different approach, focusing primarily on hazardous substances regulation. The Environmental Protection Authority (EPA) oversees cosmetic products under the Cosmetic Products Group Standard, which specifically addresses substances that may pose environmental or health risks. This regulatory distinction means brands must develop parallel compliance strategies for each market.
Key Regulatory Bodies
| Country | Primary Authority | Focus Area | Pre-Market Requirements |
|---|---|---|---|
| Australia | AICIS | Industrial chemicals notification | Mandatory for new ingredients |
| New Zealand | EPA | Hazardous substances control | Required for restricted substances |
| Both | TGA (certain products) | Therapeutic goods | Sunscreens, anti-dandruff products |
Ingredient Restrictions and Prohibited Substances
Skincare formulation standards in New Zealand and Australia include comprehensive lists of prohibited and restricted ingredients. Both countries reference international standards while maintaining unique requirements that reflect local priorities. Australia's Prohibited and Restricted Ingredients List contains over 1,400 substances with usage limitations, while New Zealand's approach identifies specific hazardous substances requiring special controls.
The recent overhaul of New Zealand's cosmetic regulations has expanded the scope of hazardous ingredient oversight, bringing the country's standards closer to European Union requirements. This regulatory evolution demonstrates the dynamic nature of skincare formulation standards and the importance of staying current with changing requirements.
Common Restricted Ingredients
- Preservatives: Parabens, formaldehyde donors, and phenoxyethanol have specific concentration limits
- Colorants: Only approved colorants from designated lists may be used in cosmetic applications
- UV filters: Sunscreen ingredients face additional restrictions and require therapeutic goods registration
- Fragrance allergens: Twenty-six allergenic fragrances must be declared when present above threshold concentrations
- Heavy metals: Strict limits on lead, mercury, arsenic, and cadmium in finished products
When developing custom formulations, ingredient selection must account for both countries' restrictions from the earliest development stages. This proactive approach prevents costly reformulation and delays in market entry.
Good Manufacturing Practice Requirements
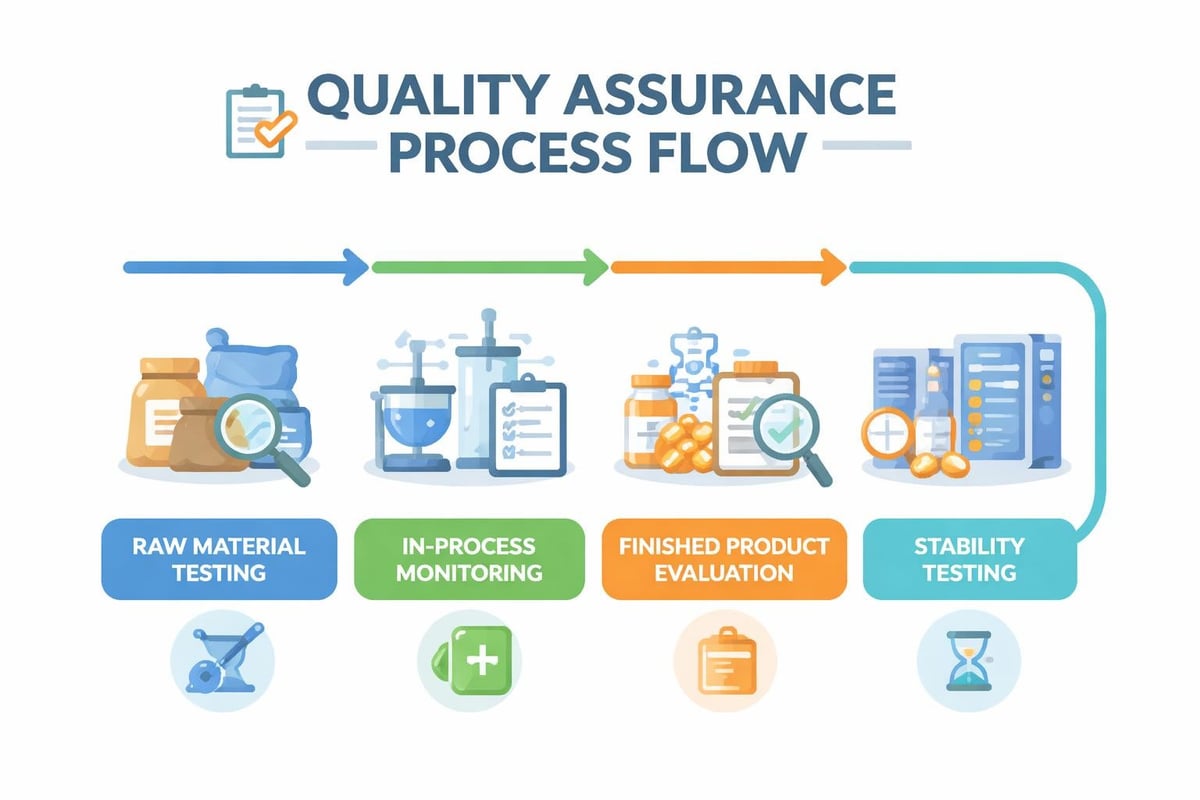
Good Manufacturing Practice (GMP) compliance represents a cornerstone of skincare formulation standards in New Zealand and Australia. While not legally mandated for all cosmetic products in New Zealand, GMP certification demonstrates commitment to quality and consumer safety. The GMP Compliance Monitoring Programme provides independent verification of manufacturing standards.
Australian regulations increasingly expect GMP adherence, particularly for brands making therapeutic claims or exporting to international markets. GMP encompasses facility design, equipment maintenance, personnel training, raw material controls, and finished product testing.
GMP Core Elements
- Documentation Systems: Batch records, standard operating procedures, and quality control documentation
- Facility Standards: Appropriate environmental controls, sanitation protocols, and segregated production areas
- Raw Material Controls: Supplier qualification, certificate of analysis verification, and quarantine procedures
- Production Processes: Validated manufacturing procedures, in-process controls, and equipment calibration
- Quality Assurance: Stability testing, microbiological testing, and complaint handling systems
- Personnel Training: Documented training programs covering hygiene, safety, and quality procedures
Understanding the role of cosmetic chemists in formulation development helps brands appreciate how technical expertise ensures GMP compliance from formulation design through production.
Labeling and Claims Requirements
Accurate labeling forms an integral component of skincare formulation standards in New Zealand and Australia. Both countries require comprehensive ingredient disclosure using the International Nomenclature of Cosmetic Ingredients (INCI) system. The ingredient list must appear in descending order of concentration, with ingredients below one percent listable in any order.
New Zealand's requirements for non-hazardous cosmetics include specific labeling obligations that extend beyond ingredient lists. Product names, net contents, manufacturer details, and warnings must meet prescribed standards. Australian regulations similarly mandate detailed labeling with additional requirements for specific product categories.
Mandatory Label Elements:
- Product name and intended use
- Ingredient list using INCI nomenclature
- Net contents by weight or volume
- Manufacturer or responsible party details
- Country of origin
- Batch code or lot number
- Special warnings for specific ingredients
- Directions for use when necessary
Claims Substantiation
Marketing claims require robust substantiation under the regulatory frameworks governing both countries. Claims about product performance, efficacy, or benefits must be truthful, not misleading, and supported by appropriate evidence. The Australian Competition and Consumer Commission actively monitors cosmetic advertising for false or deceptive representations.
| Claim Type | Evidence Required | Regulatory Scrutiny |
|---|---|---|
| Efficacy claims | Clinical studies, consumer testing | High |
| Natural/organic | Ingredient sourcing documentation | Medium |
| Hypoallergenic | Allergen testing, formulation review | High |
| Dermatologist tested | Professional evaluation records | Medium |
| Sustainable/eco-friendly | Supply chain verification | Growing |
Safety Assessment and Documentation
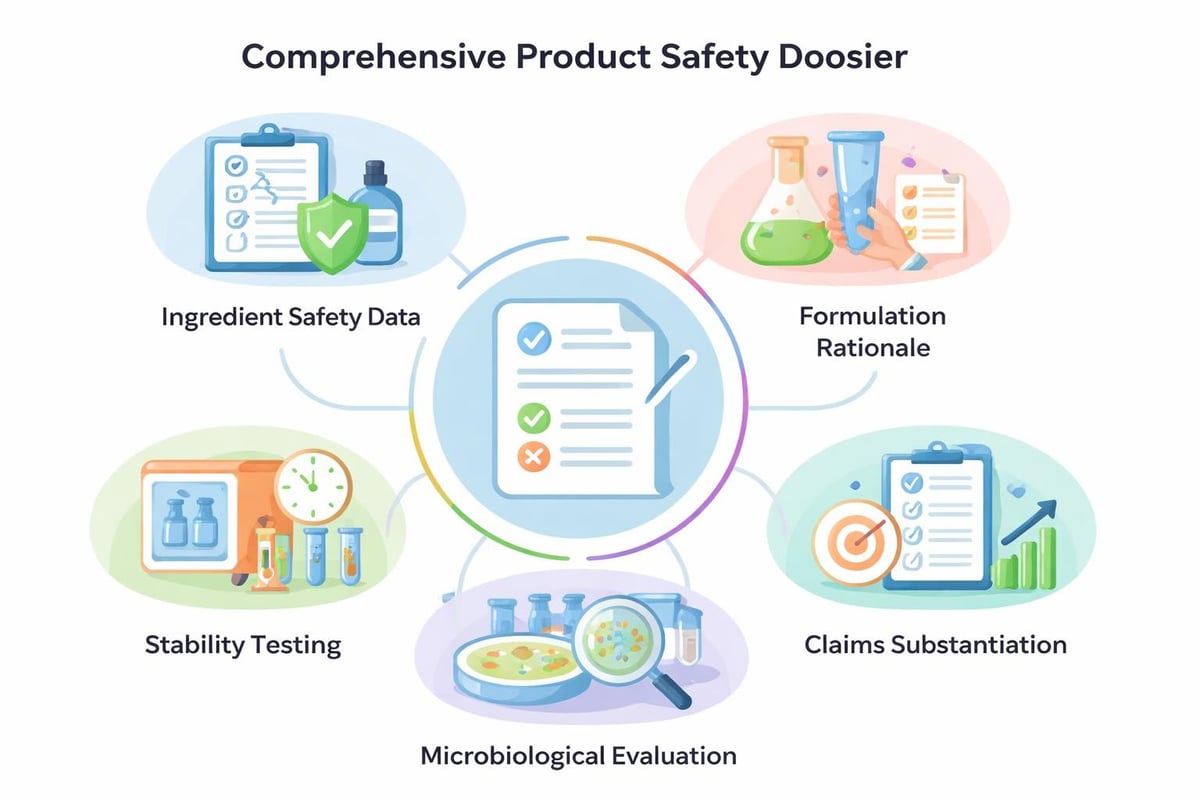
Comprehensive safety assessment underpins skincare formulation standards in New Zealand and Australia. Product Information Files (PIFs) or Cosmetic Safety Reports document the safety evaluation process, ingredient specifications, manufacturing methods, and intended use patterns. While not required for submission in all cases, maintaining thorough safety documentation enables rapid regulatory response and demonstrates due diligence.
Safety assessors evaluate ingredient safety profiles, potential interactions, exposure scenarios, and cumulative effects. This assessment considers the product's intended use, application site, duration of contact, and target demographic. Products designed for children, pregnant women, or other vulnerable populations require enhanced safety scrutiny.
The importance of quality formulations in cosmetic products extends beyond regulatory compliance to encompass brand reputation and consumer trust. Thorough safety assessment protects both consumers and brand owners from adverse events and associated liabilities.
Documentation Best Practices
- Maintain complete raw material specifications including INCI names, CAS numbers, and purity data
- Document all safety assessments with references to peer-reviewed toxicological studies
- Keep stability testing records demonstrating product integrity throughout shelf life
- Preserve microbiological challenge testing results confirming preservative efficacy
- Store batch production records with traceability to raw material lots
- Retain complaint records and adverse event reports with investigation outcomes
Therapeutic Goods and Border Categories
Certain product categories straddle the line between cosmetics and therapeutic goods under skincare formulation standards in New Zealand and Australia. Sunscreens, anti-dandruff shampoos, antiperspirants, and products making therapeutic claims fall under the Therapeutic Goods Administration (TGA) jurisdiction in Australia. These products require pre-market approval and ongoing compliance monitoring beyond standard cosmetic requirements.
New Zealand similarly regulates therapeutic claims through Medsafe, though the threshold for therapeutic classification differs from Australia. Products making claims to treat, prevent, or cure medical conditions typically require medicines classification and corresponding regulatory approval.
Borderline Product Categories:
- Sunscreens with SPF claims
- Anti-acne treatments containing active ingredients
- Anti-aging products claiming cellular repair
- Skin lightening products with restricted actives
- Products containing vitamins at therapeutic concentrations
Navigating these border categories requires careful formulation strategy and claims development. Many brands choose to formulate products as cosmetics while avoiding therapeutic claims to streamline regulatory pathways.
Sustainability and Environmental Considerations
Environmental responsibility increasingly influences skincare formulation standards in New Zealand and Australia. Both countries demonstrate strong consumer preference for sustainable, eco-friendly cosmetic brands that minimize environmental impact. While not always explicitly mandated, environmental considerations affect ingredient selection, packaging choices, and manufacturing processes.
The EPA's role in New Zealand cosmetics regulation reflects environmental priorities, with particular attention to ingredients that may accumulate in ecosystems or harm aquatic life. Microplastic restrictions, biodegradability assessments, and sustainable sourcing expectations shape formulation decisions.
Environmental Formulation Strategies
| Strategy | Implementation | Regulatory Relevance |
|---|---|---|
| Biodegradable ingredients | Select readily biodegradable surfactants and emulsifiers | EPA compliance |
| Reef-safe formulations | Avoid oxybenzone, octinoxate in sunscreen products | Market access |
| Microplastic elimination | Replace plastic microbeads with natural exfoliants | Regulatory requirement |
| Sustainable sourcing | Choose certified organic, fair trade ingredients | Claims substantiation |
| Water conservation | Develop waterless or concentrated formulations | Market differentiation |
Testing and Verification Requirements
Rigorous testing validates compliance with skincare formulation standards in New Zealand and Australia. Stability testing demonstrates product integrity under various storage conditions, typically including accelerated aging studies and real-time shelf life evaluation. Microbiological testing confirms preservative efficacy and absence of harmful pathogens.
Challenge testing subjects preserved products to deliberate microbial contamination to verify preservation system effectiveness. This testing proves particularly critical for water-based formulations susceptible to bacterial and fungal growth. Many brands conduct compatibility testing to ensure packaging materials don't interact adversely with formulation components.
Essential Testing Protocols:
- Stability Testing: 3-month accelerated and 12-month real-time studies minimum
- Microbiological Testing: Total viable count, yeast and mold, pathogens screening
- Challenge Testing: Preservative efficacy against specified organisms
- pH Testing: Verification of skin-appropriate pH ranges
- Compatibility Testing: Package-product interaction assessment
- Allergen Testing: HRIPT or RIPT for products with allergen concerns
Import and Export Considerations
For brands operating across the Tasman, understanding import requirements complements knowledge of domestic skincare formulation standards in New Zealand and Australia. Both countries require import declarations, safety documentation, and labeling compliance for cosmetic products entering commerce. The Australia New Zealand Closer Economic Relations Trade Agreement facilitates trade but doesn't eliminate regulatory requirements.
Exporters must ensure products meet destination country standards, which may differ from country of manufacture requirements. Working with contract manufacturing partners experienced in both markets streamlines compliance and reduces market entry barriers.
Import Documentation Requirements
- Commercial invoices with accurate product descriptions
- Safety Data Sheets for hazardous ingredients
- Certificates of Analysis for key ingredients
- Manufacturing certificates and GMP documentation
- Labeling compliance declarations
- Therapeutic Goods Registration (if applicable)
Quality Management Systems
Implementing comprehensive quality management systems supports ongoing compliance with skincare formulation standards in New Zealand and Australia. ISO 22716 provides internationally recognized guidelines for cosmetic GMP, offering structured frameworks for quality assurance. Many successful brands adopt quality management principles exceeding minimum regulatory requirements.
Quality systems encompass supplier qualification, change control procedures, deviation management, and continuous improvement protocols. These systems create organizational capability to maintain compliance as regulations evolve and business scales. For entrepreneurs developing cosmetic brands, establishing quality systems early prevents costly remediation later.
Quality System Components:
- Document control and record retention policies
- Supplier approval and monitoring procedures
- Deviation investigation and corrective action processes
- Internal audit schedules and finding resolution
- Management review and continuous improvement initiatives
- Training programs with competency verification
Staying Current with Regulatory Changes
The dynamic nature of skincare formulation standards in New Zealand and Australia requires ongoing regulatory monitoring. Both countries periodically update ingredient restrictions, labeling requirements, and manufacturing expectations. Recent regulatory developments include expanded hazardous substance oversight in New Zealand and increased focus on per- and polyfluoroalkyl substances (PFAS) in both countries.
Subscription to regulatory bulletins, participation in industry associations, and engagement with regulatory consultants helps brands stay informed. The regulatory landscape continues evolving in response to emerging science, consumer concerns, and international harmonization efforts.
Professional formulation partners like The INCI Lab maintain current regulatory knowledge as part of comprehensive formulation services. This expertise proves invaluable for brands lacking internal regulatory resources or entering these markets for the first time.
Navigating skincare formulation standards in New Zealand and Australia demands technical expertise, regulatory knowledge, and commitment to quality that extends throughout product development and manufacturing. The INCI Lab brings award-winning formulation capabilities and comprehensive regulatory understanding to help brands succeed in these sophisticated markets. Whether you're launching your first product or expanding an established line, The INCI Lab provides the expertise, sustainable ingredient sourcing, and full formulation ownership needed to create compliant, high-performance products that stand out in competitive beauty markets.