Understanding inci cosmetics terminology represents a fundamental requirement for anyone developing beauty products in today's market. The International Nomenclature of Cosmetic Ingredients (INCI) system provides a standardized language that formulators, manufacturers, and consumers use to identify ingredients across global markets. Whether you're launching your first skincare line or expanding an established brand, mastering INCI conventions directly impacts your product labeling, regulatory compliance, and consumer trust. This comprehensive guide explores how inci cosmetics nomenclature shapes modern formulation practices and what it means for your brand's success.
Understanding the INCI System in Modern Cosmetics
The INCI nomenclature system emerged as a solution to global confusion surrounding cosmetic ingredient identification. Before standardization, the same ingredient could appear under dozens of different names across various countries, creating regulatory challenges and consumer misunderstanding.
The International Cosmetic Ingredient Nomenclature Committee establishes these standardized names, ensuring consistency across international borders. This committee reviews ingredient submissions, assigns official INCI names, and maintains the comprehensive database that serves as the industry's authoritative reference.
Why INCI Names Matter for Your Formulation Business
When developing custom formulations, using correct inci cosmetics naming isn't optional. It's a legal requirement in most major markets, including the United States, European Union, and numerous other countries.
Key benefits of proper INCI implementation include:
- Regulatory compliance across multiple jurisdictions
- Clear communication with contract manufacturers
- Enhanced consumer transparency and trust
- Simplified ingredient sourcing and quality control
- Protection against legal challenges and product recalls
Brand owners working with formulation partners must verify that all ingredient declarations follow official INCI conventions. Even minor deviations can trigger regulatory issues that delay product launches or result in costly reformulations.
Decoding INCI Cosmetics Ingredient Lists
Reading an inci cosmetics label requires understanding the logic behind ingredient ordering and naming conventions. Ingredients must appear in descending order of predominance by weight, with those present at 1% or less allowed to appear in any order following those above 1%.
Common INCI Naming Patterns
Different ingredient categories follow specific naming conventions that formulators should recognize:
| Ingredient Type | INCI Format | Example |
|---|---|---|
| Plant Extracts | Latin botanical name + Part + Extract | Camellia Sinensis Leaf Extract |
| Chemical Compounds | Scientific chemical name | Sodium Hyaluronate |
| Mixtures | Individual components listed | Butylene Glycol, Aqua, Glycerin |
| Trade Names | INCI equivalent required | Vitamin E = Tocopherol |
Chemical ingredients typically use their scientific nomenclature, while botanical ingredients incorporate the Latin binomial name followed by the plant part. This dual approach acknowledges both traditional chemistry and botanical sciences.
The comprehensive INCI database maintained by the Personal Care Products Council contains over 30,000 entries, serving as the definitive resource for ingredient identification. Formulators should consult this database when developing new products to ensure accurate labeling.
Special Cases in INCI Nomenclature
Certain ingredients present unique challenges in inci cosmetics labeling. Fragrances, for instance, can be listed simply as "Parfum" or "Fragrance" without disclosing individual aromatic compounds, though allergens above specific thresholds must be separately identified.
Complex ingredients requiring special attention:
- Polymers: Often listed by their chemical structure rather than trade names
- Natural complexes: May require multiple INCI names for a single material
- Nanomaterials: Must include the prefix "[nano]" in the EU
- Color additives: Follow specific CI (Color Index) numbering systems
Understanding these nuances prevents labeling errors that could necessitate expensive package redesigns or regulatory interventions.
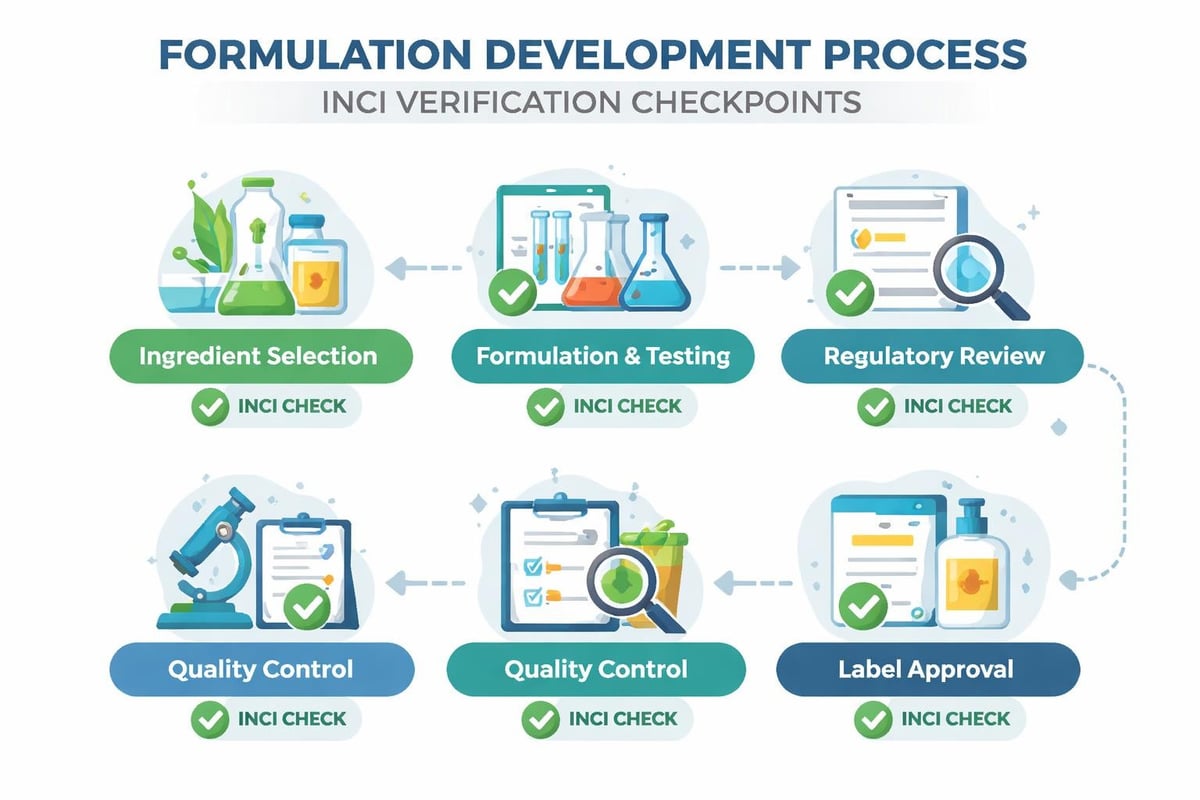
Building Compliant Formulations with INCI Standards
Creating successful personal care products requires integrating INCI compliance from the earliest formulation stages. Rather than treating ingredient naming as an afterthought during labeling design, savvy formulators build INCI considerations into their development process.
Ingredient Selection Strategy
When choosing ingredients for your formulation, verify the official INCI name before finalizing specifications. Suppliers sometimes use trade names or proprietary designations that differ from the required INCI declaration.
A systematic approach to ingredient verification includes:
- Cross-referencing supplier documentation against official INCI databases
- Requesting Certificates of Analysis with INCI names included
- Confirming that blend ingredients list all components
- Understanding regional variations in INCI naming conventions
- Maintaining updated records as INCI names occasionally change
This diligence proves especially critical when developing eco-friendly cosmetic brands where ingredient transparency drives purchasing decisions. Consumers increasingly scrutinize labels, making accuracy essential for brand credibility.
Working with Raw Material Suppliers
Establishing clear communication protocols with ingredient suppliers prevents inci cosmetics labeling mistakes. Request that all technical data sheets include official INCI names alongside trade names or product codes.
| Supplier Document | Required INCI Information |
|---|---|
| Technical Data Sheet | Primary INCI name, synonyms |
| Certificate of Analysis | INCI name matching lot number |
| Safety Data Sheet | INCI name in composition section |
| Specification Sheet | INCI name with percentage ranges |
When suppliers provide blended ingredients, demand a complete INCI breakdown of all components. A single "active complex" might contain five or more individual ingredients that must appear separately on your product label.
Regional Differences in INCI Cosmetics Regulations
While INCI provides international standardization, implementation requirements vary by jurisdiction. Understanding these regional nuances ensures your formulations meet all applicable requirements.
United States Requirements
In the U.S., the FDA requires cosmetic products to list ingredients using INCI nomenclature on the information panel. The regulatory framework mandates specific label placement and formatting, though enforcement differs from European standards.
U.S.-specific considerations include:
- Voluntary cosmetic registration program (VCRP)
- State-level requirements that may exceed federal standards
- Different allergen disclosure requirements compared to EU
- Trade secret exemptions for certain ingredients
Brands selling in multiple markets must often create region-specific labels, even when using identical formulations. This complexity underscores the importance of working with experienced formulation partners who understand international compliance.
European Union Regulations
The EU maintains stricter cosmetic regulations through its Cosmetics Regulation (EC) No 1223/2009. INCI naming must follow the European Commission's CosIng database, which serves as the official reference for the European market.
European-specific requirements extend beyond basic INCI listing to include nanomaterial disclosure, allergen identification, and specific warnings for certain ingredient categories. Products containing restricted substances must display appropriate usage instructions and warnings, all linked to specific INCI names.
Leveraging INCI Knowledge for Market Success
Sophisticated understanding of inci cosmetics nomenclature provides competitive advantages beyond mere compliance. Brands that excel at ingredient communication build stronger consumer relationships and differentiate themselves in crowded markets.
Transparent Marketing Through INCI
Modern consumers increasingly research ingredients before purchasing. Tools like INCI Beauty allow customers to analyze product compositions and evaluate ingredient safety profiles based on INCI listings.
Forward-thinking brands embrace this transparency by:
- Providing detailed ingredient education on product pages
- Explaining the purpose and benefits of each INCI component
- Highlighting sustainable or innovative ingredients by their INCI names
- Creating content that demystifies complex ingredient terminology
- Building trust through honest disclosure rather than ingredient hiding
When launching a new skincare brand, ingredient storytelling represents a powerful differentiator. Rather than viewing INCI requirements as burdensome, successful brands transform ingredient lists into compelling narratives about formulation excellence.
Competitive Analysis Using INCI
Understanding inci cosmetics labeling enables reverse-engineering competitor formulations. By analyzing ingredient order and concentration patterns, experienced formulators can estimate product compositions and identify market gaps.
This competitive intelligence supports strategic decisions about:
- Ingredient selection for unique positioning
- Cost optimization while maintaining performance
- Trend identification in emerging ingredient categories
- Regulatory compliance approaches across brands
- Claims substantiation strategies
Professional formulation partners leverage this INCI-based analysis to help brands create products that stand out while remaining compliant and cost-effective.

Common INCI Cosmetics Mistakes and How to Avoid Them
Even experienced brands occasionally stumble with inci cosmetics compliance. Recognizing common pitfalls helps prevent costly errors during product development and launch.
Incorrect Ingredient Order
One frequent mistake involves listing ingredients out of proper sequence. Remember that ingredients present above 1% must appear in descending order by weight, while those at or below 1% can appear in any order after the 1% threshold.
Red flags indicating ordering errors:
- Water-soluble actives listed before water in a cream formulation
- Preservatives appearing too early in the list for typical usage levels
- Identical products with different ingredient sequences across batches
- Fragrances listed among primary ingredients rather than near the end
Maintaining accurate batch records during formulation development prevents these ordering mistakes. Each batch sheet should document exact percentages to ensure consistent label accuracy across production runs.
Trade Name Confusion
Another common error involves using trade names instead of INCI names on labels. A supplier might market an ingredient as "SuperGlow Complex," but this proprietary name cannot appear on your INCI list without the corresponding official nomenclature.
Always verify that blended ingredients break down into individual INCI components. Your formulation might include only one bottle from your supplier, but your label might require five separate INCI names to properly declare that blend's composition.
Strategic Formulation Development with INCI Principles
Successful formulation requires thinking beyond individual ingredients to consider how INCI declarations influence consumer perception and regulatory approval. Strategic formulators balance performance, cost, and labeling impact when selecting ingredients.
Clean Beauty and INCI Transparency
The clean beauty movement has intensified consumer scrutiny of personal care ingredients. While "clean" lacks regulatory definition, many consumers evaluate products by researching INCI names against various databases and rating systems.
Brands targeting this market segment should:
- Prioritize ingredients with consumer-friendly INCI names
- Avoid ingredients commonly flagged by rating systems, even when safe
- Consider botanical extracts over synthetic alternatives when performance equals
- Invest in education around controversial but safe ingredients
- Maintain transparency about formulation choices and ingredient functions
This approach doesn't mean avoiding all synthetic ingredients or following arbitrary "free-from" lists. Rather, it requires thoughtful ingredient selection that balances efficacy with consumer comfort levels.
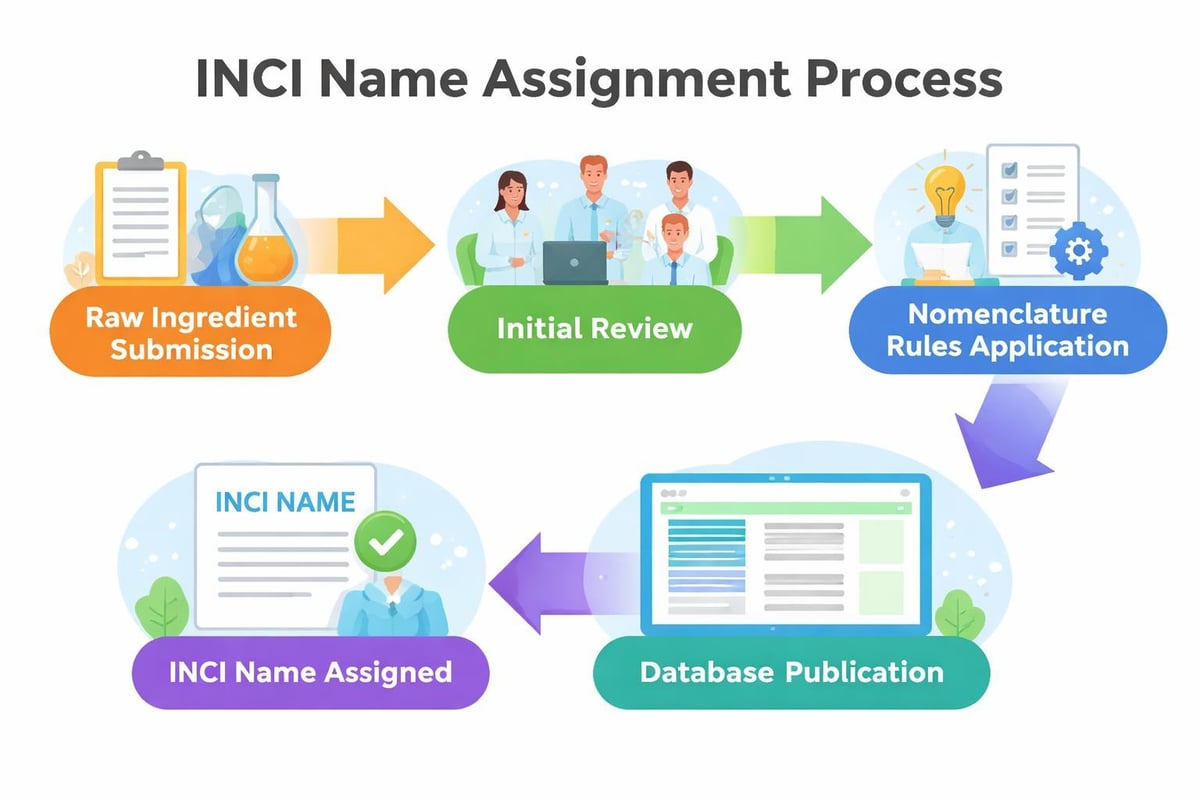
Innovation and New INCI Submissions
When developing cutting-edge formulations with novel ingredients, you might encounter materials without established INCI names. In these cases, companies must submit ingredients to the nomenclature committee for official naming.
This process involves:
- Gathering complete ingredient characterization data
- Submitting detailed chemical or botanical information
- Paying applicable fees to the nomenclature authority
- Awaiting committee review and name assignment
- Receiving the official INCI name for labeling use
The timeline for new INCI assignments varies but typically requires several months. Forward-thinking formulators account for this delay when planning product launches with innovative ingredients.
Quality Control and INCI Verification
Maintaining INCI accuracy requires robust quality control systems that verify ingredient identity throughout the supply chain. Even minor substitutions or formulation changes can invalidate existing labels and require costly reprinting.
Documentation Best Practices
Comprehensive documentation supports INCI compliance and facilitates efficient product updates. Essential records include:
| Document Type | Purpose | Update Frequency |
|---|---|---|
| Master Formula Record | Official ingredient percentages with INCI names | With each formulation change |
| Label Artwork Archive | Approved labels matching specific formula versions | With each design revision |
| Supplier Qualification | INCI verification for each raw material source | Annual review minimum |
| Batch Production Records | Actual ingredients used in specific production runs | Every batch |
This documentation proves invaluable during regulatory inspections, customer inquiries, or reformulation projects. Well-organized records enable quick verification that labels accurately reflect actual product compositions.
Managing Formulation Changes
Even minor ingredient substitutions require label updates when they affect INCI declarations. Switching from one brand of glycerin to another might not change the INCI name, but replacing a botanical extract with a different species absolutely requires label revision.
Establish clear change control procedures that:
- Require INCI review before approving any ingredient substitution
- Assess whether changes affect product labels or claims
- Update master formula records with new INCI information
- Revise labels before producing batches with changed formulations
- Maintain version control linking formulas to label artwork
These procedures prevent situations where products ship with inaccurate labels due to untracked formulation modifications.
Building Consumer Trust Through INCI Education
While inci cosmetics nomenclature serves regulatory purposes, it also represents an opportunity for consumer education and brand building. Companies that help customers understand ingredient lists foster deeper loyalty and trust.
Educational Content Strategy
Creating content that demystifies INCI terminology positions your brand as an authority while addressing consumer concerns. Effective educational approaches include:
- Ingredient glossaries explaining common INCI names in plain language
- Video content showing how to read and interpret ingredient lists
- Blog posts exploring specific ingredients and their benefits
- Social media content highlighting interesting INCI facts
- Interactive tools helping consumers find products matching their preferences
Understanding beauty INCI terminology empowers consumers to make informed choices rather than relying solely on marketing claims or influencer recommendations.
Addressing Ingredient Concerns
Some INCI names sound intimidating despite representing safe, effective ingredients. Proactive brands address these concerns by explaining why certain ingredients appear in formulations and providing scientific context.
When consumers question ingredients, respond with:
- Clear explanations of ingredient function and safety profiles
- Links to authoritative sources supporting ingredient safety
- Context about regulatory approval and testing requirements
- Transparency about why you selected specific ingredients
- Willingness to reformulate if consumer concerns reflect genuine issues
This approach builds trust even when consumers initially express skepticism about certain INCI names.
Future Trends in INCI Cosmetics
The inci cosmetics landscape continues evolving as new ingredients, technologies, and consumer expectations emerge. Staying ahead of these trends positions brands for long-term success.
Biotechnology and Novel Ingredients
Biotech-derived ingredients increasingly appear in formulations, bringing unique INCI considerations. Fermentation-derived actives, lab-grown botanicals, and bioidentical compounds require accurate nomenclature that reflects their origin and composition.
The nomenclature committee continues adapting INCI conventions to accommodate these innovations while maintaining clarity and consistency. Formulators working with cutting-edge ingredients should monitor nomenclature updates and participate in industry discussions shaping these standards.
Digital Integration and Smart Labels
Technology increasingly bridges the gap between INCI lists and consumer understanding. QR codes, NFC tags, and augmented reality features allow brands to provide detailed ingredient information beyond what fits on physical labels.
These digital enhancements enable:
- Comprehensive ingredient education accessible via smartphone
- Allergen alerts for consumers with specific sensitivities
- Sustainability information about ingredient sourcing
- Detailed explanations of complex INCI names
- Links to clinical studies supporting ingredient efficacy
Forward-thinking brands embrace these technologies to transform INCI compliance from a regulatory checkbox into a consumer engagement opportunity.
Regulatory Harmonization Efforts
Global regulatory bodies continue working toward greater harmonization of cosmetic regulations, with INCI nomenclature representing an area of strong international cooperation. Future developments may include:
- Expanded coordination between regional nomenclature authorities
- Standardized approaches to emerging ingredient categories
- Enhanced databases linking INCI names to safety assessments
- Improved processes for assigning names to novel ingredients
- Greater transparency in nomenclature decision-making
Brands operating internationally benefit from these harmonization efforts, which simplify compliance across multiple markets.
Mastering inci cosmetics nomenclature represents far more than regulatory compliance; it's a strategic advantage that enhances formulation quality, consumer trust, and market success. Whether you're developing your first product or expanding an established portfolio, partnering with formulation experts who understand the intricacies of INCI standards ensures your products meet regulatory requirements while resonating with informed consumers. The INCI Lab combines deep expertise in cosmetic nomenclature with award-winning formulation capabilities, supporting brands from initial concept through production with transparent, compliant, and high-performance products designed for competitive markets.