Understanding what is cosmetic stability testing represents a critical milestone for any brand developing personal care products, whether you're launching your first serum or expanding an established line. This systematic evaluation process determines how your formulation performs under various environmental conditions over time, ensuring customers receive products that maintain their integrity from manufacturing through the end of their shelf life. For entrepreneurs working with specialized teams who focus on quality formulations in cosmetic products, stability testing provides the scientific foundation for making reliable shelf-life claims and protecting brand reputation.
Understanding the Fundamentals of Stability Testing
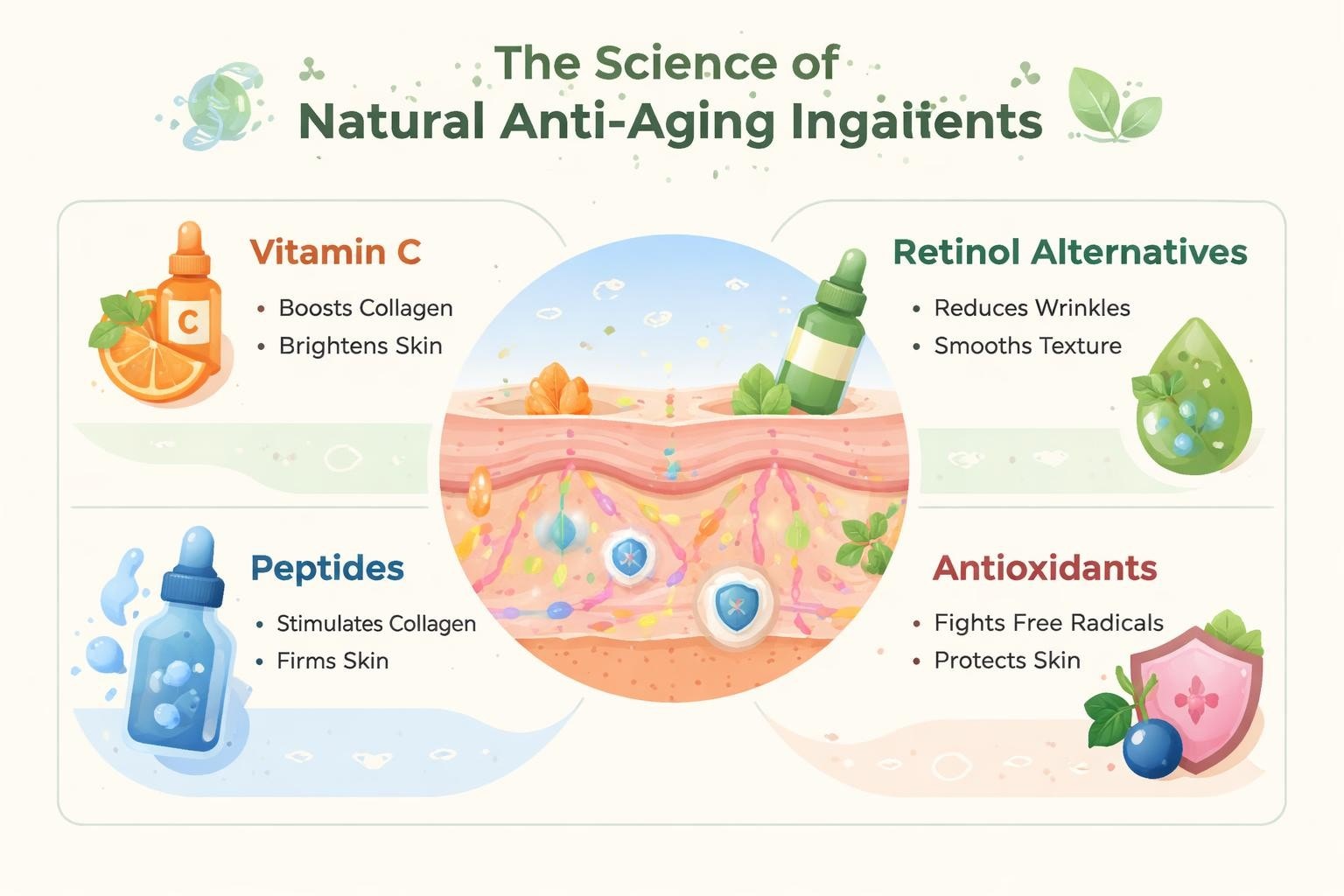
What is cosmetic stability testing at its core? It's a comprehensive evaluation protocol that examines how formulations respond to environmental stressors including temperature fluctuations, light exposure, humidity changes, and the passage of time. This testing measures physical, chemical, and microbiological stability to ensure products remain safe, effective, and aesthetically pleasing throughout their intended shelf life.
The process involves storing product samples under controlled conditions and monitoring specific parameters at predetermined intervals. Scientists evaluate changes in appearance, texture, scent, pH levels, viscosity, and active ingredient concentration. These measurements provide objective data about product degradation patterns and help determine appropriate expiration dates.
Physical Stability Evaluation
Physical stability testing monitors observable characteristics that directly impact consumer perception and product performance. Changes in color, separation of emulsions, crystal formation, or texture modifications signal potential formulation weaknesses that require adjustment before commercial launch.
Key physical parameters include:
- Color consistency across different batches and storage conditions
- Viscosity measurements to detect thickening or thinning
- Phase separation in emulsions, suspensions, and multi-phase systems
- Particle size distribution in dispersed formulations
- Package compatibility to prevent interactions with containers

Chemical Stability Assessment
Chemical stability examines whether active ingredients maintain their potency and whether the formulation develops undesirable chemical changes. This aspect proves particularly crucial for products containing sensitive ingredients like retinoids, vitamin C derivatives, or botanical extracts that can oxidize or degrade.
Testing protocols measure pH drift, oxidation markers, active ingredient assays, and the formation of degradation products. For brands working in the cosmetic industry, chemical stability data supports efficacy claims and ensures consumers receive the promised benefits throughout the product's lifespan.
Types of Stability Testing Protocols
Different testing methodologies serve distinct purposes in the product development timeline. Understanding what is cosmetic stability testing requires familiarity with these various approaches and when to apply each one.
Real-Time Stability Testing
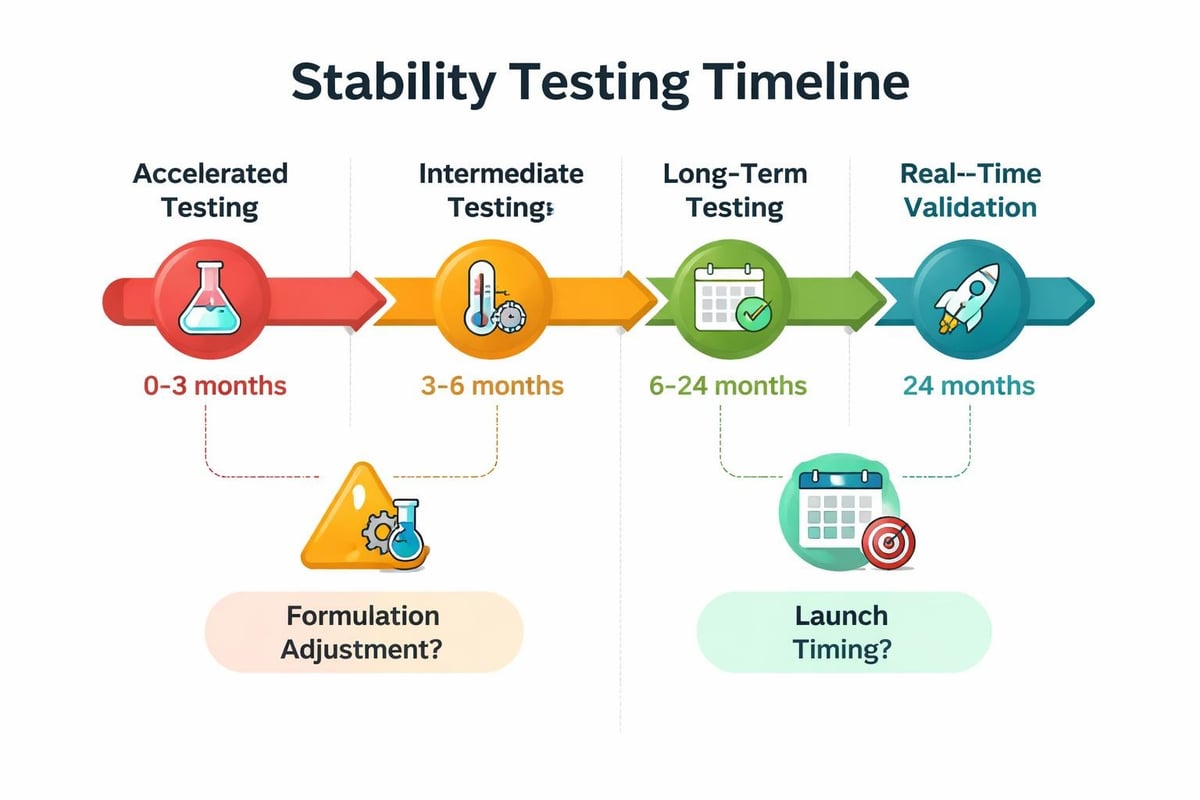
Real-time testing stores products under normal storage conditions for the entire proposed shelf life period. Samples remain at room temperature (typically 20-25°C) with standard humidity levels, mimicking actual consumer storage scenarios. This method provides the most accurate shelf-life data but requires extended timeframes.
For a product claiming a three-year shelf life, real-time testing spans 36 months with regular testing intervals at three, six, twelve, eighteen, twenty-four, and thirty-six months. MS Clinical Research explains the significance of this approach in determining accurate shelf life for cosmetic products.
Accelerated Stability Testing
Accelerated testing exposes products to elevated temperatures and humidity to simulate aging in compressed timeframes. Common conditions include 40°C with 75% relative humidity, allowing formulators to predict long-term stability within several months rather than years.
The principle behind accelerated testing involves the Arrhenius equation, which correlates temperature increases with reaction rate acceleration. Every 10°C temperature rise approximately doubles chemical reaction rates, enabling predictive modeling for extended shelf life.
| Testing Type | Temperature | Duration | Purpose |
|---|---|---|---|
| Real-Time | 20-25°C | 12-36 months | Actual shelf life validation |
| Accelerated | 40°C | 3-6 months | Predictive stability assessment |
| Intermediate | 30°C | 6-12 months | Supporting data for real-time |
| Photostability | Light exposure | Variable | Light sensitivity evaluation |
Freeze-Thaw Cycling
Freeze-thaw testing subjects formulations to repeated temperature extremes, cycling between freezing conditions and room temperature. This protocol simulates shipping conditions during winter months or storage in unheated warehouses, identifying potential emulsion breaking or ingredient precipitation.
Products typically undergo five to ten freeze-thaw cycles, with visual inspection and analytical testing conducted after completion. Formulations that survive this rigorous testing demonstrate robust stability across diverse climate zones.
Critical Parameters Monitored During Testing
What is cosmetic stability testing measuring beyond simple appearance? The comprehensive evaluation encompasses multiple analytical parameters that collectively define product quality and safety.
Microbiological Stability
Microbiological testing ensures products resist contamination throughout their shelf life, particularly important for water-based formulations. Challenge testing introduces specific microorganisms to verify preservative system effectiveness, while periodic microbial counts during stability studies confirm ongoing protection.
QACS Lab discusses various stability testing methods including microbial quality evaluation under different conditions, essential for maintaining product safety standards.
pH Monitoring
pH levels influence product safety, efficacy, and skin compatibility. Many active ingredients function optimally within specific pH ranges, and significant pH drift indicates chemical instability or ingredient degradation. Regular pH measurements throughout stability testing identify formulation adjustments needed to maintain consistent performance.
Packaging Compatibility Assessment
The container-product relationship significantly impacts stability outcomes. Testing evaluates whether packaging materials leach chemicals into the formulation, whether products stain or degrade containers, and whether closures maintain adequate seals preventing contamination or moisture loss.
Alfa Chemistry offers insights into comprehensive stability testing services, including packaging stability evaluation to ensure complete product quality.
Regulatory Compliance and Industry Standards
Understanding what is cosmetic stability testing includes recognizing regulatory expectations across different markets. While cosmetic stability testing requirements vary by region, most regulatory bodies expect manufacturers to substantiate shelf-life claims with appropriate scientific data.
FDA Expectations
The U.S. Food and Drug Administration doesn't mandate specific stability testing protocols for cosmetics but expects manufacturers to ensure product safety and proper labeling. Companies must substantiate any shelf-life claims, and stability data provides this scientific backing.
EU Cosmetics Regulation
European regulations require more formal stability testing documentation as part of the Product Information File (PIF). The EU expects manufacturers to conduct stability studies appropriate to the product type, packaging, and intended storage conditions.
ISO and Industry Guidelines
International Organization for Standardization (ISO) provides voluntary standards for cosmetic stability testing methodologies. Industry associations publish best practice guidelines that many formulators follow to ensure comprehensive evaluation protocols.
Strategic Timing for Stability Testing
Effective product development integrates stability testing at multiple stages rather than treating it as a final checkpoint. This approach saves time and resources while ensuring market-ready formulations.
Preliminary Stability Screening
Early-stage testing during formulation development identifies obvious stability issues before significant investment in full-scale studies. Brief accelerated tests lasting two to four weeks reveal major incompatibilities or instabilities, allowing rapid formulation adjustments.
For brands exploring eco-friendly cosmetic formulations, preliminary screening proves particularly valuable given the unique stability challenges some natural ingredients present.
Pre-Market Stability Studies
Before commercial launch, comprehensive stability testing validates that finalized formulations meet quality standards throughout their proposed shelf life. This stage includes accelerated testing to support initial shelf-life estimates and begins the real-time testing program that continues post-launch.
Post-Market Surveillance
Ongoing stability monitoring of marketed products confirms that manufacturing scale-up hasn't introduced stability concerns and that products continue meeting quality standards. This surveillance supports brand reputation and consumer safety.
Common Stability Challenges and Solutions
What is cosmetic stability testing revealing about frequent formulation problems? Certain issues appear consistently across product categories, each requiring specific strategic solutions.
Emulsion Separation
Creams and lotions may separate into distinct phases due to inadequate emulsifier systems, improper manufacturing processes, or environmental stress. Solutions include optimizing emulsifier selection, adjusting manufacturing parameters, or incorporating stabilizing polymers.
Color Changes
Oxidation, ingredient interactions, or light exposure cause color shifts that alarm consumers even when products remain safe and effective. Antioxidants, chelating agents, opaque packaging, and careful ingredient selection minimize color instability.
Microbial Growth
Inadequate preservation allows bacterial or fungal contamination, particularly dangerous in products applied near eyes or on broken skin. Broad-spectrum preservative systems, proper manufacturing hygiene, and careful pH control maintain microbiological stability.
| Stability Issue | Common Causes | Potential Solutions |
|---|---|---|
| Phase separation | Inadequate emulsifiers | Optimize surfactant system |
| Oxidation | Air exposure, light | Add antioxidants, use airless packaging |
| pH drift | Ingredient reactions | Buffer systems, ingredient substitution |
| Viscosity loss | High temperatures | Thickener adjustment, polymer networks |
Specialized Testing for Specific Product Categories
Different cosmetic products face unique stability challenges requiring tailored testing approaches. Understanding what is cosmetic stability testing for your specific product type ensures appropriate protocol selection.
Sunscreen Formulations
Photostability testing proves critical for sunscreen products, ensuring UV filters maintain their protective properties despite light exposure. Certified Laboratories details stability testing services including photostability tests that support shelf-life and efficacy claims.
Natural and Organic Products
Formulations emphasizing botanical ingredients or excluding synthetic preservatives require especially rigorous stability testing. Natural extracts may oxidize, change color, or develop off-odors more readily than synthetic alternatives. Working with experienced formulators who understand contract manufacturing for beauty products helps navigate these challenges.
Products with Active Ingredients
Serums, treatments, and therapeutic products containing active ingredients like peptides, enzymes, or vitamins need stability testing that monitors active potency throughout shelf life. Degradation of active ingredients compromises product efficacy and consumer trust.

Interpreting Stability Testing Results
Raw stability data requires expert interpretation to inform formulation decisions and shelf-life determinations. What is cosmetic stability testing telling you about your product's future performance?
Acceptance Criteria
Establishing clear acceptance criteria before testing begins provides objective standards for evaluating results. These criteria specify acceptable ranges for each measured parameter, defining when changes constitute product failure versus normal variation.
Typical acceptance criteria might include:
- Visual appearance remains consistent with initial product
- pH changes stay within ±0.5 units of original value
- Active ingredient content maintains 90-110% of label claim
- Microbial counts remain below specified limits
- Viscosity changes stay within ±20% of original measurement
Failure Investigation
When products fail stability testing, systematic investigation identifies root causes and guides reformulation efforts. Changes to preservative systems, antioxidant packages, emulsifier blends, or manufacturing processes may resolve stability issues without compromising product performance or brand identity.
Documentation and Record-Keeping
Comprehensive documentation proves essential for regulatory compliance, quality assurance, and continuous improvement. Stability testing generates extensive data that requires organized management and long-term retention.
Stability Protocol Development
Written protocols specify testing conditions, sampling schedules, analytical methods, and acceptance criteria before studies begin. These documents ensure consistency across multiple product batches and testing cycles, enabling meaningful comparisons and trend analysis.
ISCA Cosmetic Testing outlines the process of stability testing for cosmetic products, detailing parameters evaluated and documentation importance for ensuring product safety and compliance.
Data Management Systems
Organized data systems track test results, facilitate trend analysis, and support regulatory submissions. Many companies implement laboratory information management systems (LIMS) that integrate stability data with other quality control information.
Cost Considerations and ROI
Understanding what is cosmetic stability testing financially involves balancing upfront testing costs against potential market failures, recalls, or reputation damage. While comprehensive testing requires investment, it proves far less expensive than launching unstable products.
Testing Investment Breakdown
Stability testing costs vary based on product complexity, testing duration, and analytical methods required. Accelerated testing provides relatively affordable preliminary data, while comprehensive three-year real-time studies represent significant ongoing commitments.
Budget considerations include:
- Laboratory fees for analytical testing and storage
- Sample preparation and shipping costs
- Specialized testing for photostability or packaging compatibility
- Extended timeline costs for real-time validation
- Reformulation expenses if initial formulations fail
Protecting Brand Investment
For entrepreneurs and established brands, stability testing protects much larger investments in product development, marketing, packaging design, and brand building. A product failure due to inadequate stability testing can damage customer relationships built over years, making comprehensive evaluation essential risk management.
Leveraging Stability Data for Marketing
Successfully completed stability testing provides valuable marketing assets beyond simple compliance documentation. What is cosmetic stability testing contributing to your brand story and consumer confidence?
Shelf-Life Claims
Scientifically validated shelf-life claims demonstrate product quality and professional development processes. Specific expiration dates or period-after-opening (PAO) symbols backed by stability data build consumer trust and differentiate professional brands from amateur competitors.
Quality Messaging
Brands can communicate their commitment to quality through references to rigorous testing programs, stability-validated formulations, and science-backed product development. This messaging particularly resonates with informed consumers who research product quality before purchasing.
Future Trends in Stability Testing
The cosmetic stability testing field continues evolving with technological advances and changing market demands. Emerging trends include accelerated analytical methods, predictive modeling using artificial intelligence, and sustainability-focused testing protocols that minimize sample waste and energy consumption.
Green chemistry principles influence testing methodology development, with researchers seeking alternatives to traditional accelerated conditions that consume significant energy for temperature control. Predictive models based on ingredient properties and formulation characteristics may eventually supplement or reduce extensive physical testing requirements.
Cosmetic stability testing represents an essential investment in product quality, consumer safety, and brand reputation, providing the scientific foundation for successful product launches and sustained market presence. Whether you're developing your first formulation or expanding an established line, The INCI Lab offers comprehensive support throughout the product development journey, from initial concept through stability-validated formulations ready for production. With award-winning expertise in custom formulations for cosmetics, personal care, and pet care products, The INCI Lab combines scientific rigor with sustainable practices to create high-performance products that stand the test of time and market scrutiny.